katie raffaini
Overview
For this project, we were asked to create our own "science fair" project based off of one or more of the concepts that we have learned this year.
After given this assignment, my partner Paige and I began to contemplate which projects were our favorites this year. We both enjoyed the
physics of sports project, as well as the musical band project. We tried to think of projects that we could create that would fall under either of
these categories, but we had no luck. We then thought about our last project, which was electricity and robot art. We thought about the circuits
that we created and how much we learned from that project. Then we thought about the idea of potato clocks. After doing some research, we `
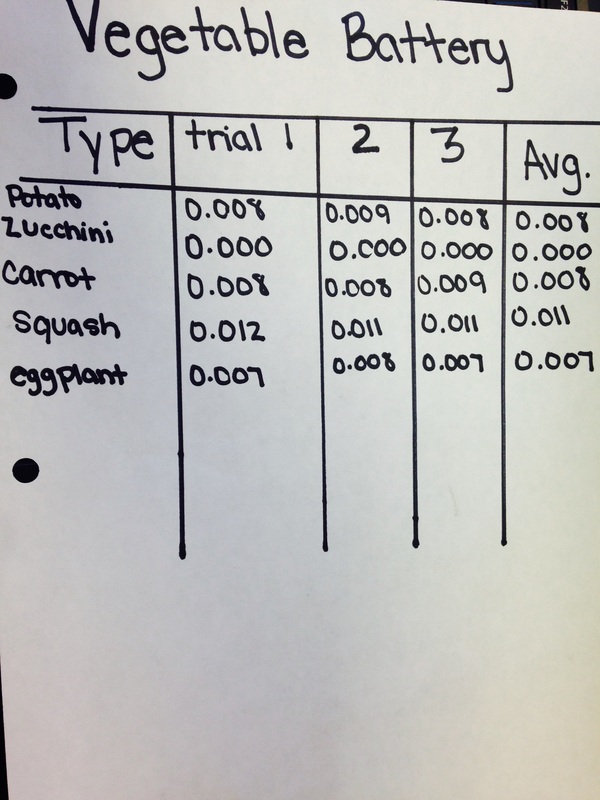
came up with the idea of vegetable batteries. We bought a potato, carrot, squash, zucchini, and an eggplant. We used each one to create a
circuit. Our goal was to find which out of our five vegetables could be used as a battery, and which ones worked the best.
Our first day at work on this project, we just went over what we were going to do for this project and which vegetables we were going to
use. On the second day, we did more research. On the third day, we watched videos about how to use potatoes as batteries and we replicated
it. The next day, we had the rest of our batteries, so we were able to make the rest of the batteries. Unfortunately, vegetables go bad over time,
so we weren't able to preserve them to present in class on the last day of school. However, we did get pictures.
Here are our results:
After given this assignment, my partner Paige and I began to contemplate which projects were our favorites this year. We both enjoyed the
physics of sports project, as well as the musical band project. We tried to think of projects that we could create that would fall under either of
these categories, but we had no luck. We then thought about our last project, which was electricity and robot art. We thought about the circuits
that we created and how much we learned from that project. Then we thought about the idea of potato clocks. After doing some research, we `
came up with the idea of vegetable batteries. We bought a potato, carrot, squash, zucchini, and an eggplant. We used each one to create a
circuit. Our goal was to find which out of our five vegetables could be used as a battery, and which ones worked the best.
Our first day at work on this project, we just went over what we were going to do for this project and which vegetables we were going to
use. On the second day, we did more research. On the third day, we watched videos about how to use potatoes as batteries and we replicated
it. The next day, we had the rest of our batteries, so we were able to make the rest of the batteries. Unfortunately, vegetables go bad over time,
so we weren't able to preserve them to present in class on the last day of school. However, we did get pictures.
Here are our results:
How does it work?
Vegetable batteries use the acids in the vegetable to start a reaction with two electrodes made of different metals that cause electrons to flow
from one to the other through the potato, producing power. The potato acts as a salt bridge, connecting the anode, or negative electrode, to the
cathode, or positive electrode. The vegetable is not a source of electricity by itself. The potato itself has a mixture of starches and salts and
some water. A salt, like table salt, in water releases ions. Ions are atoms that have an electrical charge. Table salt releases two electrically
charged ions - a sodium ion with a positive charge, and a chlorine ion with a negative charge.To make a battery with a potato, you need to put
two different metals into the potato...the metals can be zinc and tin, or copper and steel, or other choices. The point is - the two metals have to
be different metals. You might use a penny (made of copper) and a nickel.
from one to the other through the potato, producing power. The potato acts as a salt bridge, connecting the anode, or negative electrode, to the
cathode, or positive electrode. The vegetable is not a source of electricity by itself. The potato itself has a mixture of starches and salts and
some water. A salt, like table salt, in water releases ions. Ions are atoms that have an electrical charge. Table salt releases two electrically
charged ions - a sodium ion with a positive charge, and a chlorine ion with a negative charge.To make a battery with a potato, you need to put
two different metals into the potato...the metals can be zinc and tin, or copper and steel, or other choices. The point is - the two metals have to
be different metals. You might use a penny (made of copper) and a nickel.
